Tag: generic drugs - Page 2
Planning for Patent Expiry: What Patients and Healthcare Systems Need to Do Now

Patent expirations are triggering massive drug price drops-but most patients and health systems aren’t ready. Learn what you need to do now to avoid disruptions, save money, and stay safe.
- December 7 2025
- Tony Newman
- 9 Comments
Bioequivalence Studies: What the FDA Requires Generic Drug Manufacturers to Prove
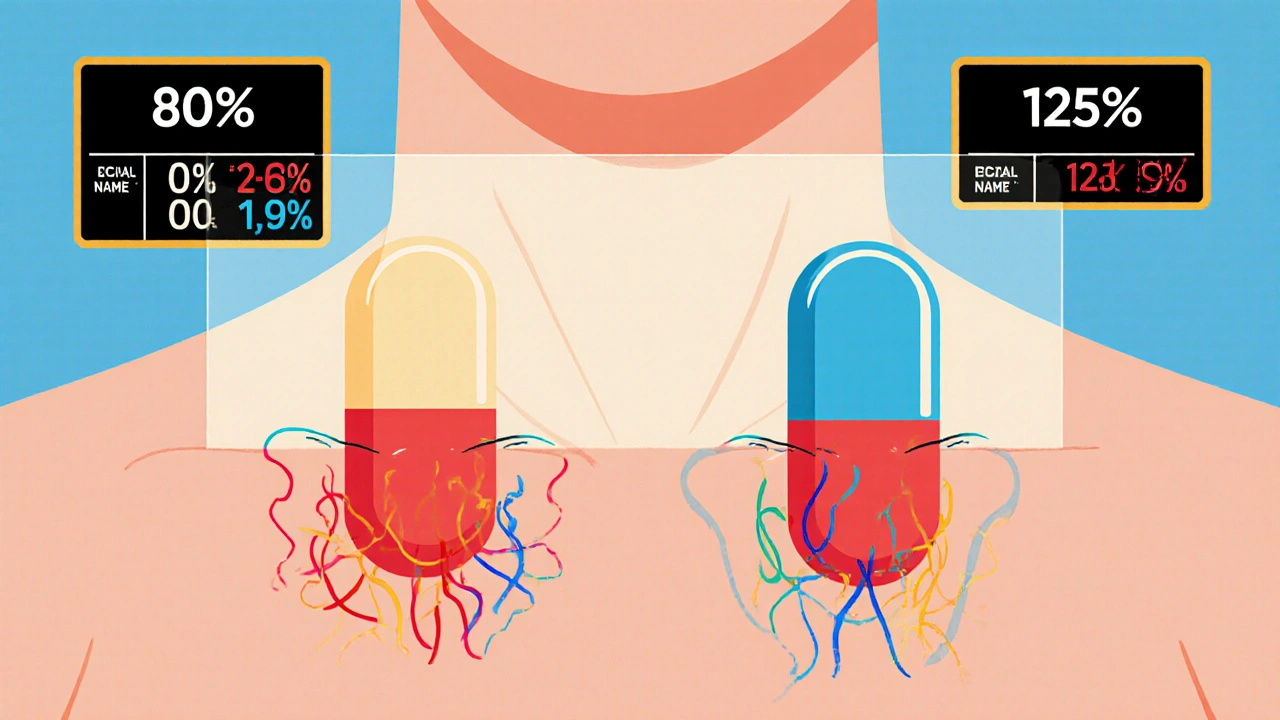
The FDA requires generic drug manufacturers to prove bioequivalence through rigorous studies showing their product absorbs at the same rate and extent as the brand-name drug. Learn the 80/125 rule, biowaivers, NTID exceptions, and why this matters for patient safety.
- November 27 2025
- Tony Newman
- 12 Comments
Single-Source vs Multi-Source Drugs: What Patients Need to Know About Cost, Effectiveness, and Choices

Understand the difference between single-source and multi-source drugs to save money and make smarter choices. Learn how pricing, generics, and insurance affect your out-of-pocket costs.
- November 21 2025
- Tony Newman
- 17 Comments
