Generic Drugs: What They Are, How They Work, and Why They Matter
When you hear generic drugs, medications that contain the same active ingredients as brand-name pills but are sold without a brand name. Also known as generic medicine, they work the same way, in the same dose, and with the same risks and benefits as their branded cousins—except they cost a fraction of the price. Most people assume brand-name drugs are better, but that’s not true. The FDA requires generic drugs to meet the same strict standards for quality, strength, purity, and performance. If your doctor prescribes a brand-name drug, you’re often getting the exact same medicine—just without the marketing budget attached.
Why does this matter? Because drug cost savings, the financial relief that comes from choosing generics instead of brand-name versions can mean the difference between taking your medicine every day or skipping doses because it’s too expensive. For people managing chronic conditions like high blood pressure, diabetes, or depression, switching to a generic can make treatment sustainable. And it’s not just about price. Many of the same safety concerns that apply to brand-name drugs—like interactions with other meds, side effects, or timing of doses—also apply to generics. That’s why understanding medication safety, the practice of using drugs correctly to avoid harm, errors, or dangerous reactions is just as important with generics as it is with brands. You still need to know what you’re taking, what to watch for, and when to call your doctor.
What you’ll find in this collection isn’t just a list of cheap pills. It’s a real-world guide to how generics fit into everyday health. You’ll read about how specialty pharmacies handle generic versions of complex drugs, how to spot when a side effect is from your medicine and not your disease, and how older adults can safely cut back on unnecessary pills—including generics that may no longer be needed. You’ll see how herbal supplements like Ginkgo Biloba can interact with blood thinners, whether evening primrose oil affects seizure risk, and how the FDA is changing how medication info is presented to patients. These aren’t theoretical discussions. They’re stories from people managing real conditions, making real choices, and living with real consequences.
Whether you’re paying out of pocket, on Medicare, or helping a parent navigate their medicine cabinet, the truth is simple: generic drugs aren’t second-rate—they’re the smart, safe, and standard choice for millions. But knowing how to use them right? That’s where the real value lies. Below, you’ll find practical, no-fluff advice on how to get the most from your prescriptions, avoid hidden risks, and take control of your health without overpaying.
Contamination Controls: How Generic Drug Manufacturers Prevent Adulteration

Contamination controls in generic drug manufacturing prevent dangerous adulteration through strict cleanroom standards, real-time monitoring, and human process discipline. Learn how facilities keep pills safe despite tight margins.
- March 4 2026
- Tony Newman
- 5 Comments
Generics vs Brands: How Insurance Coverage Policies Differ

Insurance policies treat generics and brand-name drugs very differently-even when they're medically identical. Learn how formularies, copays, prior authorizations, and state laws shape what you pay and what you get.
- March 2 2026
- Tony Newman
- 6 Comments
The Orange Book: How Therapeutic Equivalence Guides Generic Drug Substitution

The Orange Book is the FDA's official guide to therapeutic equivalence, determining which generic drugs can safely replace brand-name medications. It saves billions annually and is used daily by pharmacists, insurers, and regulators.
- February 7 2026
- Tony Newman
- 15 Comments
How Multiple Generic Drug Manufacturers Lower Prices: Real Data & Patient Tips

Learn how multiple generic drug manufacturers drive down prices through competition. Real data shows savings of up to 70% with four or more competitors. Understand how this affects your healthcare costs.
- February 6 2026
- Tony Newman
- 9 Comments
Medicare Part D Formularies: How Generic Drug Coverage Works in 2025

Medicare Part D generic drug coverage in 2025 is simpler and cheaper than ever, with a $2,000 out-of-pocket cap and low-tier copays. Learn how formularies work, what you pay, and how to save on your prescriptions.
- January 27 2026
- Tony Newman
- 10 Comments
How to Talk to Your Doctor About Staying on a Brand Medication

Learn how to talk to your doctor about staying on a brand medication when generics are available. Get practical tips, what to say, how to prepare, and how to handle insurance hurdles.
- January 17 2026
- Tony Newman
- 8 Comments
Medication Therapy Management: How Pharmacists Optimize Generic Drug Use for Better Outcomes

Pharmacists use Medication Therapy Management to help patients save money by switching to safe, effective generic drugs-cutting costs by up to 85% while improving adherence and reducing hospital visits.
- January 14 2026
- Tony Newman
- 9 Comments
How to Navigate Pharmacy Substitution Laws in Your State: Generic vs Brand Medications
Learn how pharmacy substitution laws vary by state when switching from brand-name drugs to generics or biosimilars. Know your rights, what pharmacists must do, and how to protect your health.
- January 12 2026
- Tony Newman
- 14 Comments
Planning for Patent Expiry: What Patients and Healthcare Systems Need to Do Now

Patent expirations are triggering massive drug price drops-but most patients and health systems aren’t ready. Learn what you need to do now to avoid disruptions, save money, and stay safe.
- December 7 2025
- Tony Newman
- 9 Comments
Bioequivalence Studies: What the FDA Requires Generic Drug Manufacturers to Prove
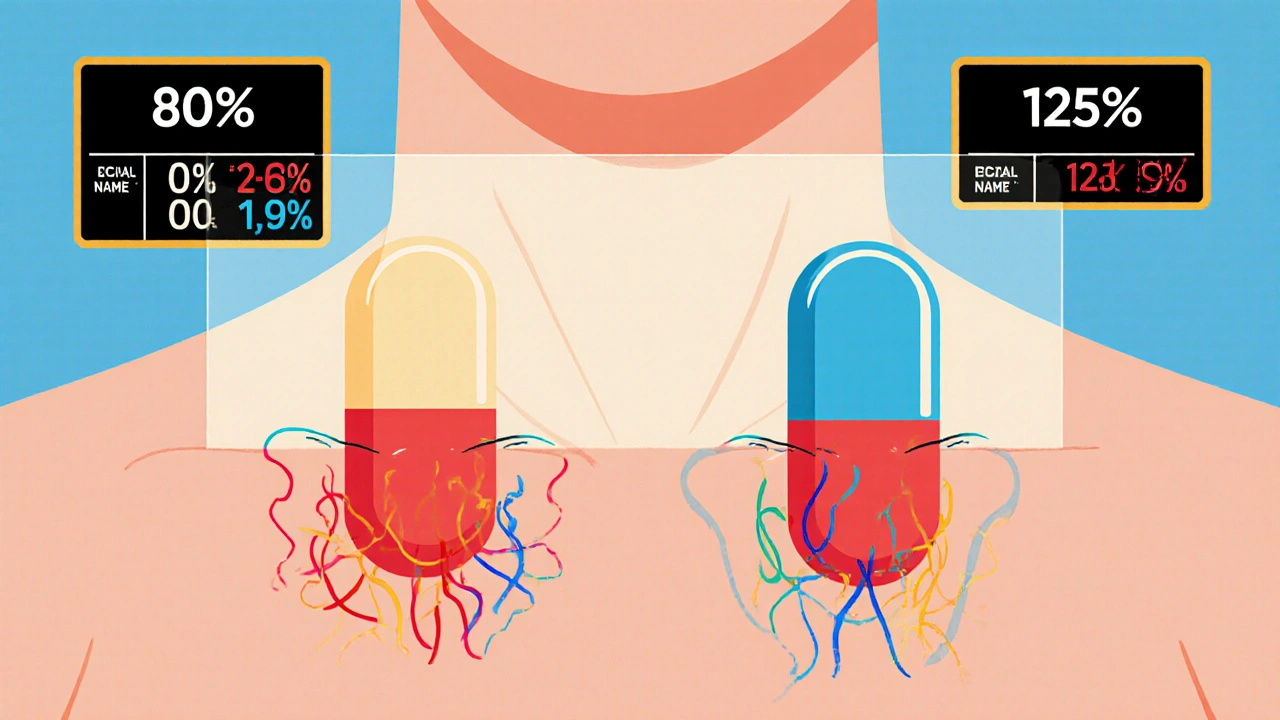
The FDA requires generic drug manufacturers to prove bioequivalence through rigorous studies showing their product absorbs at the same rate and extent as the brand-name drug. Learn the 80/125 rule, biowaivers, NTID exceptions, and why this matters for patient safety.
- November 27 2025
- Tony Newman
- 12 Comments
